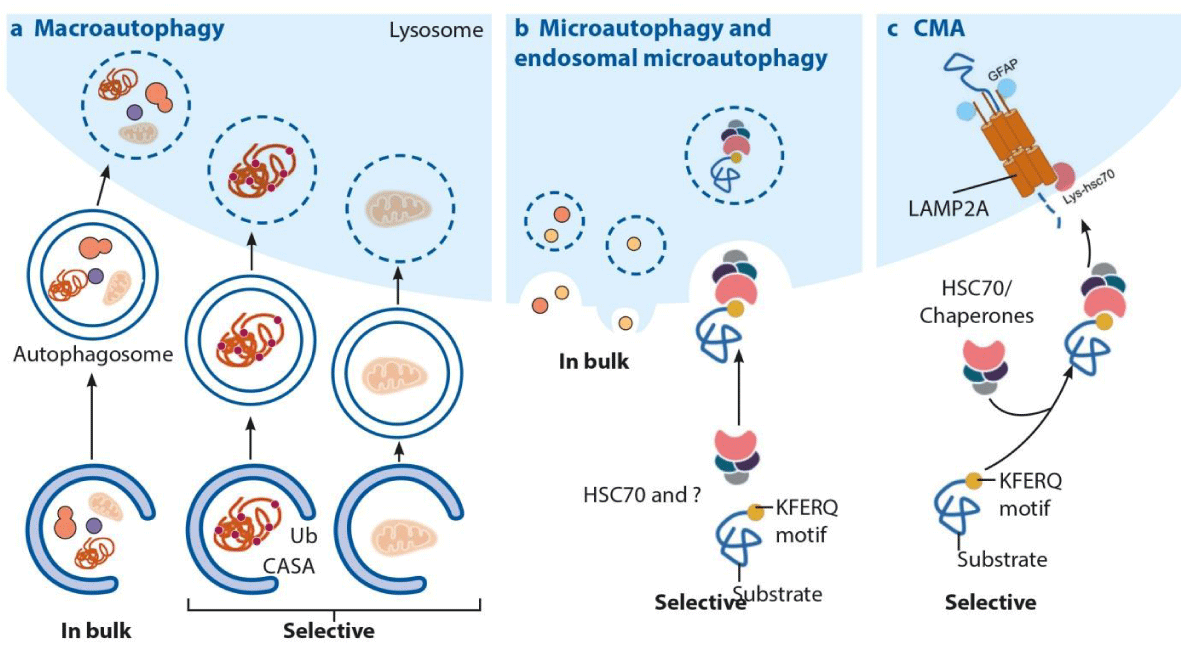
Thus, an increase in other protein degradation pathways may partially compensate for the loss of CMA in osteoblasts. Moreover, similar to what has been observed in other cell types, macroautophagy and proteasomal degradation were upregulated in CMA-deficient osteoblasts in culture. Consistent with these findings, specific knockdown of Lamp2a in an osteoblastic cell line increased RANKL expression and decreased mineral deposition. The low bone mass of L2ACgKO mice was associated with increased osteoclastogenesis and decreased mineral deposition in cultured cells. At 18 weeks of age, both male and female L2ACgKO mice had lower vertebral cancellous bone mass compared to WT controls. The low bone mass of L2ACgKO mice was associated with elevated RANKL expression and the osteoclast marker genes Trap and Cathepsin K. At 5 weeks of age female L2ACgKO mice had lower vertebral cancellous bone mass compared to wild-type (WT) controls, whereas there was no difference between genotypes in male mice at this age. CRISPR-Cas9-based genome editing led to the deletion of both Lamp2a and Lamp2c, another Lamp2 isoform, producing Lamp2AC global knockout (L2ACgKO) mice. To address the role of CMA for skeletal homeostasis, we deleted an essential component of the CMA process, namely Lamp2a, from the mouse genome. However, the role of CMA in skeletal homeostasis under physiological and pathophysiological conditions is unknown. Chaperone-mediated autophagy (CMA) is a protein degradation pathway that eliminates soluble cytoplasmic proteins that are damaged, incorrectly folded, or targeted for selective proteome remodeling.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
